Article of the Week: Safety, reliability and accuracy of small renal tumour biopsies: results from a multi-institution registry
Every week the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an accompanying editorial written by a prominent member of the urological community. This blog is intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
Safety, reliability and accuracy of small renal tumour biopsies: results from a multi-institution registry
Abstract
Objective
To validate, in a multi-institution review, the safety, accuracy and reliability of renal tumour biopsy (RTB) and its role in decreasing unnecessary treatment.
Materials and Methods
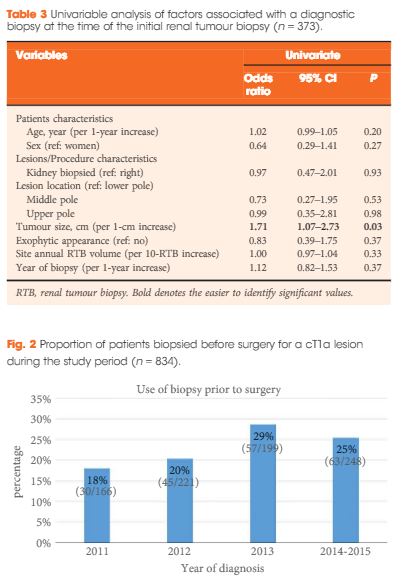
We conducted a multi-institution retrospective study of patients who underwent RTB to characterize a small renal mass (SRM) between 2011 and May 2015. Patients were identified using the prospectively maintained Canadian Kidney Cancer information system. Diagnostic and concordance rates were presented using proportions, whereas factors associated with a diagnostic RTB were identified using a logistic regression model.
Results
Of the 373 biopsied SRMs, the initial biopsy was diagnostic in 87% of cases. Of the 47 non-diagnostic biopsies, 15 had a repeat biopsy of which, 80% were diagnostic. When both were combined, therefore, a diagnosis was obtained in 91% of SRMs. Of these, 18% were benign. Size was the only factor found to be associated with achieving a diagnostic biopsy. RTB histology and nuclear grade (high or low) were found to be highly concordant with surgical pathology (86 and 81%, respectively). Of the discordant tumours (n = 16), all were upgraded from low to high grade on surgical pathology. Adverse events were rare (<1% of cases).
Conclusion
The present multi-institution study confirms that RTB of SRMs is safe, accurate and reliable across institutions, while decreasing unnecessary treatment. Given our findings, RTBs may be a helpful tool with which to triage SRMs and guide appropriate management.