Article of the week: Does the introduction of prostate multi-parametric MRI into the AS protocol for localized PCa improve patient re-classification?
Every week, the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an accompanying editorial written by a prominent member of the urological community. This blog is intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
Does the introduction of prostate multiparametric magnetic resonance imaging into the active surveillance protocol for localized prostate cancer improve patient re-classification?
Richard J. Bryant*† , Bob Yang* , Yiannis Philippou*, Karla Lam*, Maureen Obiakor*, Jennifer Ayers*, Virginia Chiocchia†‡, Fergus Gleeson§, Ruth MacPherson§, Clare Verrill†¶, Prasanna Sooriakumaran†**, Freddie C. Hamdy*† and Simon F. Brewster*
*Department of Urology, Oxford University Hospitals NHS Foundation Trust, Oxford, UK, †Nuffield Department of Surgical Sciences, University of Oxford, Oxford, UK, ‡National Perinatal Epidemiology Unit, Nuffield Department of Population Health, University of Oxford, Oxford, UK, §Department of Radiology, Oxford University Hospitals NHS Foundation Trust, Oxford, UK, ¶Oxford NIHR Biomedical Research Centre, University of Oxford, Oxford, UK, and **Department of Uro-Oncology, University College London Hospital NHS Foundation Trust, London, UK
Abstract
Objectives
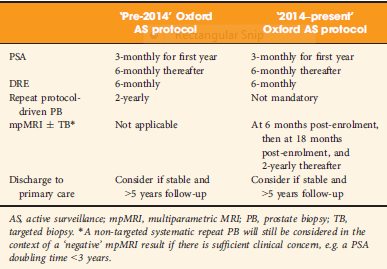
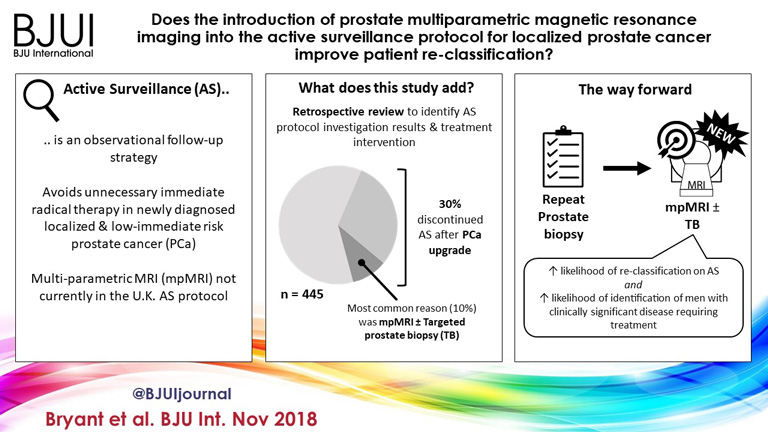
To determine whether replacement of protocol‐driven repeat prostate biopsy (PB) with multiparametric magnetic resonance imaging (mpMRI) ± repeat targeted prostate biopsy (TB) when evaluating men on active surveillance (AS) for low‐volume, low‐ to intermediate‐risk prostate cancer (PCa) altered the likelihood of or time to treatment, or reduced the number of repeat biopsies required to trigger treatment.
Patients and Methods
A total of 445 patients underwent AS in the period 2010–2016 at our institution, with a median (interquartile range [IQR]) follow‐up of 2.4 (1.2–3.7) years. Up to 2014, patients followed a ‘pre‐2014’ AS protocol, which incorporated PB, and subsequently, according to the 2014 National Institute for Health and Care Excellence (NICE) guidelines, patients followed a ‘2014–present’ AS protocol that included mpMRI. We identified four groups of patients within the cohort: ‘no mpMRI and no PB’; ‘PB alone’; ‘mpMRI ± TB’; and ‘PB and mpMRI ± TB’. Kaplan–Meier plots and log‐rank tests were used to compare groups.
Results
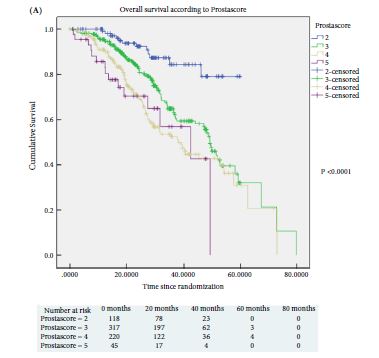
Of 445 patients, 132 (30%) discontinued AS and underwent treatment intervention, with a median (IQR) time to treatment of 1.55 (0.71–2.4) years. The commonest trigger for treatment was PCa upgrading after mpMRI and TB (43/132 patients, 29%). No significant difference was observed in the time at which patients receiving a PB alone or receiving mpMRI ± TB discontinued AS to undergo treatment (median 1.9 vs 1.33 years; P = 0.747). Considering only those patients who underwent repeat biopsy, a greater proportion of patients receiving TB after mpMRI discontinued AS compared with those receiving PB alone (29/66 [44%] vs 32/87 [37%]; P = 0.003). On average, a single set of repeat biopsies was needed to trigger treatment regardless of whether this was a PB or TB.
Conclusion
Replacing a systematic PB with mpMRI ±TB as part of an AS protocol increased the likelihood of re‐classifying patients on AS and identifying men with clinically significant disease requiring treatment. mpMRI ±TB as part of AS thereby represents a significant advance in the oncological safety of the AS protocol.