Article of the month: Three‐dimensional virtual imaging of renal tumours: a new tool to improve the accuracy of nephrometry scores
Every month, the Editor-in-Chief selects an Article of the Month from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an editorial written by a prominent member of the urology community and a video prepared by the authors; we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this week, it should be this one.
Three‐dimensional virtual imaging of renal tumours: a new tool to improve the accuracy of nephrometry scores
Abstract
Objectives
To apply the standard PADUA and RENAL nephrometry score variables to three‐dimensional (3D) virtual models (VMs) produced from standard bi‐dimensional imaging, thereby creating 3D‐based (PADUA and RENAL) nephrometry scores/categories for the reclassification of the surgical complexity of renal masses, and to compare the new 3D nephrometry score/category with the standard 2D‐based nephrometry score/category, in order to evaluate their predictive role for postoperative complications.
Materials and Methods
All patients with localized renal tumours scheduled for minimally invasive partial nephrectomy (PN) between September 2016 and September 2018 underwent 3D and 2D nephrometry score/category assessments preoperatively. After nephrometry score/category evaluation, all the patients underwent surgery. Chi‐squared tests were used to evaluate the individual patients’ grouping on the basis of the imaging tool (3D VMs and 2D imaging) used to assess the nephrometry score/category, while Cohen’s κ coefficient was used to test the concordance between classifications. Receiver‐operating characteristic curves were produced to evaluate the sensitivity and specificity of the 3D nephrometry score/category vs the 2D nephrometry score/category in predicting the occurrence of postoperative complications. A general linear model was used to perform multivariable analyses to identify predictors of overall and major postoperative complications.
Results
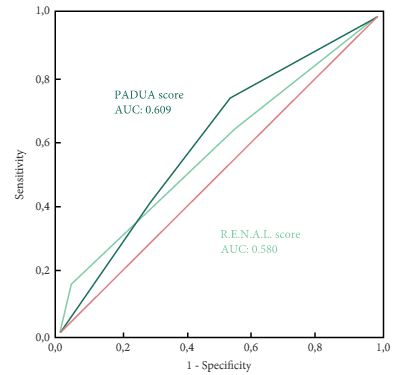
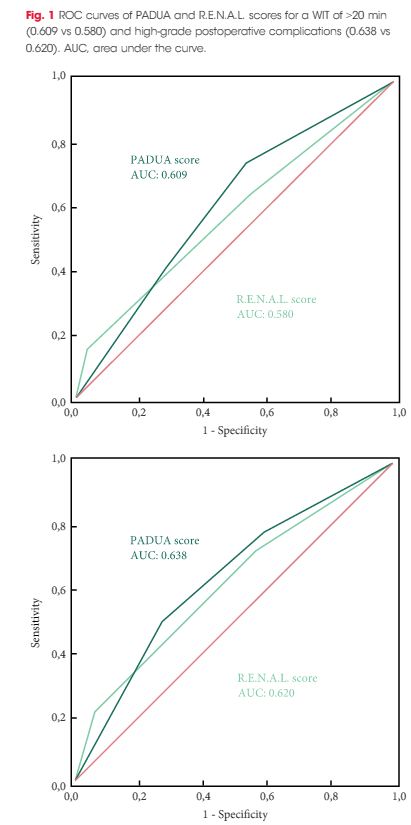
A total of 101 patients were included in the study. The evaluation of PADUA and RENAL nephrometry scores via 3D VMs showed a downgrading in comparison with the same scores evaluated with 2D imaging in 48.5% and 52.4% of the cases. Similar results were obtained for nephrometry categories (29.7% and 30.7% for PADUA risk and RENAL complexity categories, respectively). The 3D nephrometry score/category demonstrated better accuracy than the 2D nephrometry score/category in predicting overall and major postoperative complications (differences in areas under the curve for each nephrometry score/category were statistically significant comparing the 3D VMs with 2D imaging assessment). Multivariable analyses confirmed 3D PADUA/RENAL nephrometry category as the only independent predictors of overall (P = 0.007; P = 0.003) and major postoperative complications (P = 0.03; P = 0.003).
Conclusions
In the present study, we showed that 3D VMs were more precise than 2D standard imaging in evaluating the surgical complexity of renal masses according to nephrometry score/category. This was attributable to a better perception of tumour depth and its relationships with intrarenal structures using the 3D VM, as confirmed by the higher accuracy of the 3D VM in predicting postoperative complications.