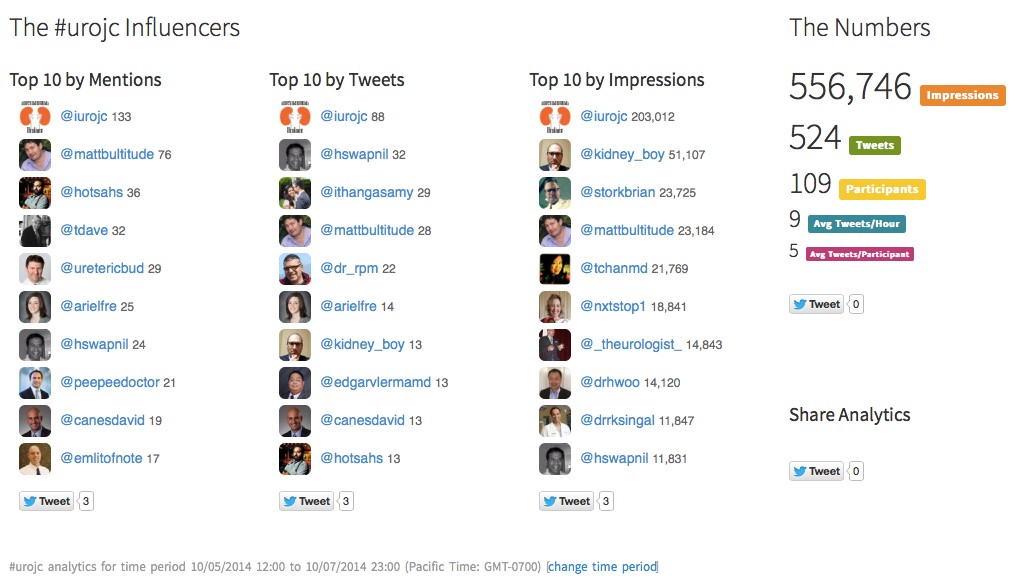
We present an unreported complication of a second-degree burn sustained by a 62-year-old male who underwent shockwave lithotripsy for two renal calculi.
Authors: Sriram Rangarajan1, Hossein Mirheydar1, Roger L. Sur1,2
1. UC San Diego Health Sciences, Department of Surgery, Division of Urology; San Diego, CA
2. VA San Diego Medical Center, San Diego, CA
Corresponding Author: Roger L. Sur, MD, UC San Diego Health Sciences, Division of Urology, 200 W Arbor Dr. #8897, San Diego, CA 92103-8897. T: 619-543-2630 E-mail: rlsur@ucsd.edu
Abstract
We present an unreported complication of a second-degree burn sustained by a 62-year-old male who underwent shockwave lithotripsy for two renal calculi. The patient was treated conservatively for his burn injury but did require several ancillary procedures to render him stone free. We speculate that the number of shocks and bubbles associated with an unapproved coupling medium for this particular lithotripter may have led to this highly unusual adverse event.
Introduction
Shockwave lithotripsy is considered a minimally invasive procedure for upper tract urinary stone disease with the obvious benefit of minimal side effects and rare adverse events. These advantages have helped evolve it into a common surgical treatment modality especially due to its safety profile. We report an unusual adverse event following shockwave lithotripsy involving a second degree skin burn at the coupling site.
Case Report
A 62-year-old Caucasian male presented with gross hematuria and left-flank pain. He had been seen in the emergency department 18 months prior for left-flank pain associated with two calcifications (a 9 mm midcalyceal calculus and an 8 mm lower pole calculus). The patient’s past medical history was unremarkable. His medications included allopurinol, 300 mg, and aspirin, 81 mg, both of which he had discontinued one week prior to procedure. Physical examination revealed mild left costovertebral angle tenderness. Computerized tomography (CT) urogram of the abdomen and pelvis demonstrated the two previously noted renal calculi (10 mm,1500 HU in the left calyx and 9mm,1000 HU in the left calyx ). Cystoscopy and cytological examination of two urine specimens did not reveal any abnormality.
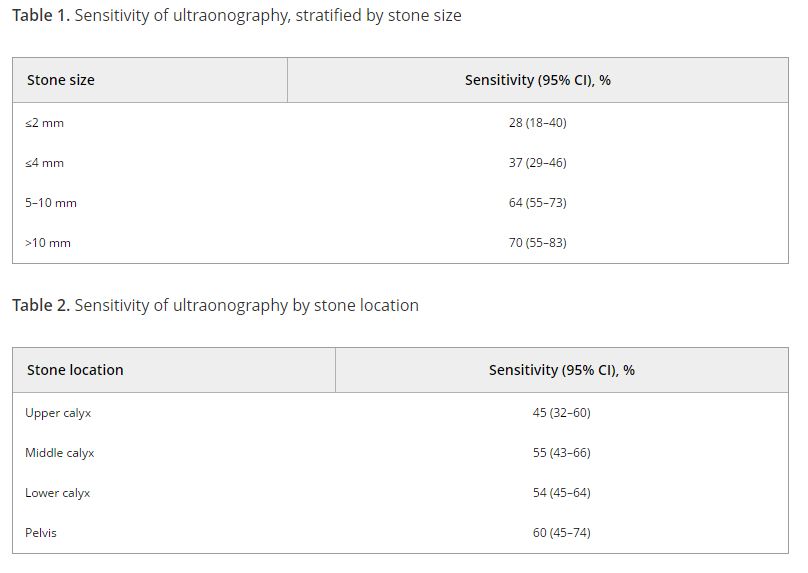
The patient opted for shockwave lithotripsy (SWL). An electromagnetic lithotripter (Medispec EM1000, Germantown, MD), which our institution leases from Medispec, was used, and 4000 shocks (2000 to each calculus) were delivered at 90 shocks/minute after gradually increasing the power to 20kV. The Medispec technician did not have the standard coupling gel recommended by Medispec and instead used ultrasound gel available from the hospital. In the recovery unit, the entire skin overlying the area where the gel had been placed was noted to be moderately erythematous with 3-4 cm central area of subcutaneous hemorrhage (figure 1).
Figure 1. demonstrates the immediate post-operative changes noted in the recovery unit: erythema and central region of petechiae
The patient returned on postoperative day 1 with significantly increased erythema and the development of large blisters (figure 2). He also noted some abdominal pain, gross hematuria and passage of small fragments. The burn surgery service was consulted. A second-degree burn was confirmed, and local debridement was performed along with application of Collegnase, polymyxin B, xeroform, and gauze dressings.
Figure 2 demonstrates development of marked erythema and bullae on post-operative day 1
The patient returned again on postoperative day 5 complaining of left-flank pain and vomiting. The wound remained erythematous with denuded skin. A noncontrast CT of his abdomen and pelvis CT demonstrated an interval decrease in stone burden but multiple fragmented calculi (900HU) in the lower calyx as well as at least two adjacent 6 mm calculi in the proximal ureter (HU 1000). The patient consented to ureteroscopy with Ho:YAG laser lithotripsy. During the left ureteroscopy with Ho:YAG laser lithotripsy and ureteric stenting, the ureteric calculi migrated into the kidney and one large calculus was fragmented. A significant amount of mucous material in the urine precluded good visualization. The indwelling stent was removed 6 days later at outpatient cystoscopy.
Renal ultrasound performed 5 weeks later demonstrated a moderate left hydronephrosis and CT of the abdomen and pelvis revealed a 2 cm column of distal ureteral calculi. Subsequent ureteroscopy was aborted because of an iatrogenic extraperitoneal bladder perforation during dilation of an unexpected bulbar urethral stricture encountered during initial cystoscopy. After documenting healing of the bladder, left ureteroscopy with Ho:YAG laser lithotripsy and ureteraic stenting was performed 10 days later. This rendered the patient stone free, as confirmed by direct visualization. Outpatient cystoscopic stent removal was performed 5 days later. He was asymptomatic two months afterwards with normal voiding and a normal appearance on renal ultrasound.
Discussion
Complications following SWL are rare but include renal hematoma 1-13 % [1-3], steinstrasse 2-10% [4], acute renal injury, and theoretically, chronic renal injury such as a risk of hypertension [5]. To our knowledge this is the first case documenting a second-degree skin burn following SWL treatment. There is no obvious explanation for this unusual adverse event, but some facts specific to this individual case deserve mention. Firstly, we acknowledge that 4000 shocks might be considered marginally high. However, this has been routinely delivered with this particular lithotriptor in appropriate cases where calculi were in different renal locations, and this adverse event had never been experienced previously. The two stones was located in different regions of the kidney and required 2000 shocks to adequately fragment under fluoroscopy. It is tempting to surmise that a fewer shocks would have prevented this outcome.
We also note that conventional ultrasound gel was used in lieu of standard coupling gel approved for use with the Medispec EM1000 lithotripter. The Medispec-recommended coupling medium is Lithoclear gel (Sonotech, Bellingham, WA). It has a density of 1.013 g/cc and is made of water-based couplants with a general composition of >80% water, 5 to 15% propylene glycol, 1 to 5 % glycerine, an acrylic polymer and a cosmetic-grade preservative. The Lithoclear gel is filled and reportedly packaged under tighter controls than ultrasound gel to ensure that the containers are absolutely free of micro- and macrobubbles that would cause reflection or scattering of shock wave acoustics during SWL [6]. Anecdotally, it has higher viscosity than ultrasound gel. Higher viscosity may result in lower tendency to form bubbles [7]. The ideal medium would mimic the viscosity of water to minimize loss of energy but would be viscous enough that it would not run off the patient during the procedure. The medium should also be devoid of air bubbles, as they are thought to further attenuate the energy transmission to the patient. Just as cavitation functions to fragment calculi during lithotripsy, some have proposed that cavitation of bubbles within the coupling medium is responsible for skin pain [7]. Could the skin burn be attributed to cavitation of excessive bubbles within the nonapproved ultrasound gel? Ultrasound gel is routinely used as a coupling agent in some centers, though ensuring the presence of bubbles is minimized is commonly advocated [8]. We can only speculate that excessive cavitation from the coupling medium associated with an elevated number of delivered shocks may have been sufficient to generate a thermal injury. Another explanation is heat generated from the water-filled cushion, though there is no plausible reason to think this is possible. The specific lithotripter used for this case was evaluated by the maker, Medispec, and no mechanical abnormalities were discovered.
References
1. Evan AP, Willis LR (2007). Extracorporeal shock wave lithotripsy: complications. In: Smith AD, Badlani G, Bagley D, eds. Smith’s Textbook of Endourology. 2nd ed. Hamilton, ON, BC Decker, 2007; chap 41: 353–65.
2. Orozco Farinas R, Iglesias Prieto JI, Massarrah Halabi J, Mancebo Gómez JM, Perez-Castro Ellendt E. Renal hematoma after extracorporeal shockwave lithotripsy in a series of 324 consecutive sessions with the DOLI-S lithotripter: incidents, characteristics, multifactorial analysis and review. Arch Espan Urol. 2008; 61: 889–914.
3. Mobley TB. Low energy lithotripsy with the lithostar: treatment results with 19,962 renal and ureteral stones. J Urol. 1993; 149: 1419–24.
4. Madbouly K, Sheir KZ, Elsobky E, Eraky I, Kenawy M. Risk factors for the formation of a steinstrasse after extracorporeal shock wave lithotripsy: a statistical model. J Urol. 2002; 167: 1239–42.
5. Lingeman JE, McAteer JA, Assimos DG, Baxley J, Kahn RI, Krambeck A, Matlaga BR, Penson D, Preminger GM, Zhong P. White Paper: Current Perspective on Adverse Effects in Shock Wave Lithotripsy. American Urological Association Education and Research, 2009.
6. Email communication with Sonotech.
7. Heidenreich A, Bonfig R, Wilbert DM, Engelmann UH. Painless ESWL by cutaneous administration of Vaseline. Urologe A. 1995 Jul;34(4):343-7. German
8. Cartledge JJ, CrossWR, Lloyd SN, Joyce AD. The efficacy of a range of contact media as coupling agents in extracorporeal shockwave lithotripsy. BJU Int. 2001; 88: 321–4.
Date added to bjui.org: 25/04/2012
DOI: 10.1002/BJUIw-2012-006-web