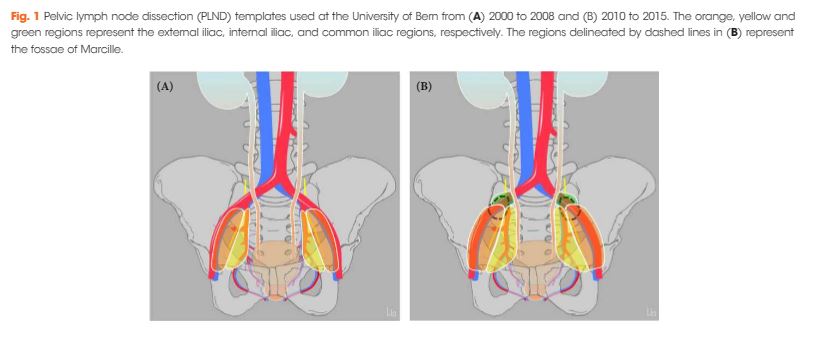
We describe a case of an isolated bladder metastasis in EWS, which, to our knowledge, has not been previously reported.
Authors: Alexander Yeates MBBS, Peter Campbell FRACS, Queen Elizabeth II Jubilee Hospital, Brisbane, Australia
Corresponding Author: Alexander Yeates MBBS, Queen Elizabeth II Jubilee Hospital, Brisbane, Australia. Email: asyeates@gmail.com, alexander_yeates@health.qld.gov.au
Abstract
Ewing’s sarcoma (EWS) can occur in almost any bone or soft tissue, however cases involving the bladder are exceedingly rare. We describe a case of an isolated bladder metastasis in EWS, which, to our knowledge, has not been previously reported.
A twelve year old boy was initially diagnosed with primary EWS of the skull. He was treated with local surgical resection, radiotherapy and chemotherapy. He had no recurrence until four years later when he presented with painless haematuria. Urinary cytology revealed small, round atypical cells, and he was referred for a urological opinion. An intravenous pyelogram was normal. Rigid cystoscopy revealed a 20 x 15mm lesion that was resected and histology confirmed recurrence of EWS. After careful consideration, a partial cystectomy was performed with good post-operative recovery and subsequently he completed a course of adjuvant chemotherapy. 40 months of follow-up including blood tests, rigid cystoscopy, CXR, CT and USS have not revealed further recurrence.
The prognosis following recurrence of Ewing’s sarcoma is usually guarded, however features specific to this case, such as the prolonged interval to recurrence and presence of a distant, isolated recurrence are relatively reassuring.
Introduction
Ewing’s sarcoma (EWS) was initially described by James Ewing in 1921 as an undifferentiated, small, round cell tumour involving the diaphysis of long bones [1]. More recently, EWS, primitive neuroectodermal tumour and Askin’s tumour have all been classified under the common term of the Ewing’s sarcoma family of tumours following identification of the common translocation t(11;22)(q24;q12) resulting in the formation of the EWS-ETS fusion gene [2,3]. These tumours can occur in almost any bone or soft tissue, however cases of EWS involving the bladder are exceedingly rare [4,5] and a case of an isolated recurrence in the bladder has not previously been reported.
Case Report
An otherwise healthy twelve year old boy was diagnosed with EWS of the skull in 2003. He was initially treated with local excision, radiotherapy (50.4Gy) and chemotherapy (vincristine 15mg/m2, cyclophosphamide 17,400mg/m2, doxorubicin 480mg/m2, ifosfamide 39,000mg/m2, carboplatin 3,000mg/m2 and etoposide 1,950mg/m2) followed by stem cell rescue. Four years later he presented to an Accident and Emergency Department following a single episode of frank haematuria. Abdominal and pelvic ultrasound scan demonstrated normal upper urinary tracts with a large amount of echogenic material consistent with clot within the bladder. Urine microscopy showed clusters of small round atypical cells, lymphocytes and erythrocytes. An intravenous pyelogram was normal.
Following referral to a Urologist, rigid cystoscopy identified a 20x15mm solid necrotic lesion on the left lateral wall of the bladder (Figure 1).
Figure 1.Rigid cystoscopy showing bladder lesion.
The lesion was excised with a 26 Fr resectoscope. Microscopic examination of the lesion revealed sheets of small, undifferentiated malignant cells invading into muscle (Figure 2).
Figure 2. Undifferentiated malignant tumour of bladder. (H&E stained section, original magnification x400)
Immunohistochemical testing showed the cells were strongly positive for CD99 in a membranous pattern with scattered cells positive for NFP and synaptophysin. They were negative for S100, Desmin, Myo D1 and SMA. Fluorescence in-situ hybridisation (FISH) using the Vysis [6] EWSR1 (22q12) dual colour break apart rearrangement probe showed rearrangement of the EWSR1 gene region confirming the diagnosis of metastatic EWS (Figure 3).
Figure 3. Fluorescence in-situ hybridisation of the bladder lesion using the Vysis EWRS1 (22q12) dual break apart probe shows rearrangement of the EWSR1 gene region, confirming the diagnosis of metastatic EWS.
Staging investigations (whole body bone scan and CT abdomen and pelvis) showed no evidence of disease elsewhere in the body.
Given that this appeared to represent an isolated tumour recurrence, the decision was made to perform a partial cystectomy in October 2007. The lesion was identified and excised with a four centimetre margin (Figure 4).
Figure 4. Partial cystectomy specimen showing central area of ulceration.
Histology of the excised specimen showed an area of ulceration that was clear of the margins. There was no evidence of residual tumour. Post-operative recovery was uneventful.
Following partial cystectomy, the patient received adjuvant chemotherapy of topotecan (36mg/m2) and cyclophosphamide (18,000mg/m2). This was adequately tolerated. The patient was considered for sperm storage but was found to be azoospermic.
Follow-up has involved surveillance rigid cystoscopy and examination under anaesthesia at three-monthly intervals with interim outpatient reviews with surveillance full blood count, electrolytes and liver function blood tests, chest x-ray, abdominal and pelvic CT and ultrasound scans to exclude further metastatic disease. Surveillance for over 40 months has so far revealed no evidence of local recurrence or further metastases.
Discussion
Rare cases of primary EWS of the bladder have been reported [4,5] and EWS has also been described as arising in the bladder as a second tumour in paediatric patients with a previous haematological malignancy [7]. To our knowledge this case represents the first report of primary skeletal EWS with an isolated bladder metastasis. Given that the five-year survival rate for patients with recurrent EWS has been reported to be as low as 13% [8], the initial prognosis for this patient was guarded. Significant risk factors for death following recurrence in Leavey’s study [9] included recurrence at combined local and distant sites, elevated LDH at initial diagnosis and initial recurrence less than two years following diagnosis. In contrast, this patient had recurrence at a single distant site four years after the primary diagnosis of EWS and is now disease free four years after excision of the recurrent lesion. This is relatively reassuring when considering the long-term prognosis.
It is also worth noting that this adolescent patient presented clinically with an episode of macroscopic haematuria. This symptom cannot be ignored and should lead to consideration of cystoscopic examination of the bladder particularly in patients with a significant previous medical history [10].
References
1. Ewing J. Diffuse endothelioma of bone. Proc NY Pathol Soc 1921;21:17-24.
2. Aurias A, Rimbaut C, Buffe D, Zucker JM, Mazabraud A. Translocation involving chromosome 22 in Ewing’s sarcoma: a cytogenic study of four fresh tumors. Cancer Genet Cytogenet 1984;12:21-25.
3. Whang-Peng J, Triche TJ, Knutsen T, Miser J, Douglass EC, Israel MA. Chromosomal translocation in peripheral neuroepithelioma. N Engl J Med 1984;311:584-585.
4. Gousse AE, Roth DR, Popek EJ, Cooley LD, Horowitz ME. Primary Ewing’s sarcoma of the bladder associated with an elevated antinuclear antibody titer. J. Urol. 1997;158:2265-2266.
5. Okada Y, Kamata S, Akashi T, Kurata M, Nakamura T, Kihara K. Primitive neuroectodermal tumor/Ewing’s sarcoma of the urinary bladder: a case report and its molecular diagnosis. Int J Clin Oncol. 2010 Nov 10. [Epub ahead of print].
6. Vysis package insert. Invitrogen Spot-Light tissue Pre-treatment Kit package insert. KOJI,T, Molecular Histochemical Techniques, Springer- Verlag, Tokyo, 2000.
7. Osone S, Hosoi H, Tanaka K, Tsuchiya K, Iehara T, Morimoto A, Hashida T, Yamashita M, Kawabata K, Nishijo K, Toguchida J, Hata J, Sugimoto T. A case of a Ewing sarcoma family tumor in the urinary bladder after treatment for acute lymphoblastic leukemia. J Pediatr Hematol Oncol. 2007 Dec;29(12):841-4.
8. Bacci G, Ferrari S, Longhi A, Donati D, De Paolis M, Forni C, Versari M, Setola E, Briccoli A, Barbieri E. Therapy and survival after recurrence of Ewing’s tumours: the Rizzoli experience in 195 patients treated with adjuvant and neo-adjuvant chemotherapy from 1979 to 1997. Ann. Oncol 2003;14:1654-1659.
9. Leavey PJ, Mascarenhas L, Marina N, Chen Z, Krailo M, Miser J, Brown K, Tarbell N, Bernstein ML, Granowetter L, Gebhardt M, Grier HE. Prognostic Factors for Patients with Ewing sarcoma (EWS) at First Recurrence Following Multimodality Therapy – A Report from the Children’s Oncology Group. Pediatr Blood Cancer. 2008 September ; 51(3): 334–338.
10. Gordon C, Stapleton FB. Hematuria in adolescents. Adolesc Med Clin. 2005;16:229-39.
Date added to bjui.org: 06/09/2011
DOI: 10.1002/BJUIw-2011-028-web