Article of the week: The effect of the urinary and faecal microbiota on lower urinary tract symptoms measured by the International Prostate Symptom Score: analysis utilising next‐generation sequencing
Every week, the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
Please use the comment buttons below if you would like to join the conversation.
If you only have time to read one article this week, we recommend this one.
The effect of the urinary and faecal microbiota on lower urinary tract symptoms measured by the International Prostate Symptom Score: analysis utilising next‐generation sequencing
Bradley Holland*, Mallory Karr*, Kristin Delfino*, Danuta Dynda†, Ahmed El-Zawahry‡, Andrea Braundmeier-Fleming*, Kevin McVary§ and Shaheen Alanee¶
*Southern Illinois University School of Medicine, †Center for Clinical Research, Southern Illinois University School of Medicine, Springfield, IL, USA, ‡Urology, University of Toledo, Toledo, OH, §Loyola University Chicago, Chicago, IL, and ¶Vattikuti Urology Institute, Henry Ford Hospital, Detroit, MI, USA
Abstract
Objective
To examine the correlation between urinary and faecal microbial profiles and the different aspects of lower urinary tract symptoms (LUTS) in men, as there is accumulating evidence that variations in the human microbiota may promote different benign disease conditions.
Patients and Methods
We extracted total DNA from urine and faecal samples of a group of men, under an Institutional Review Board‐approved protocol. At the same time, International Prostate Symptom Score (IPSS) data were collected. We then amplified the extracted DNA and sequenced it using bacterial 16S ribosomal RNA gene high‐throughput next‐generation sequencing platform, and analysed the microbial profiles for taxonomy to examine the correlation between the different operational taxonomy units (OTUs) and LUTS represented by the total IPSS, the different symptom levels of the IPSS (mild, moderate, and severe) and its subcomponents of storage, nocturia, voiding, and bother.
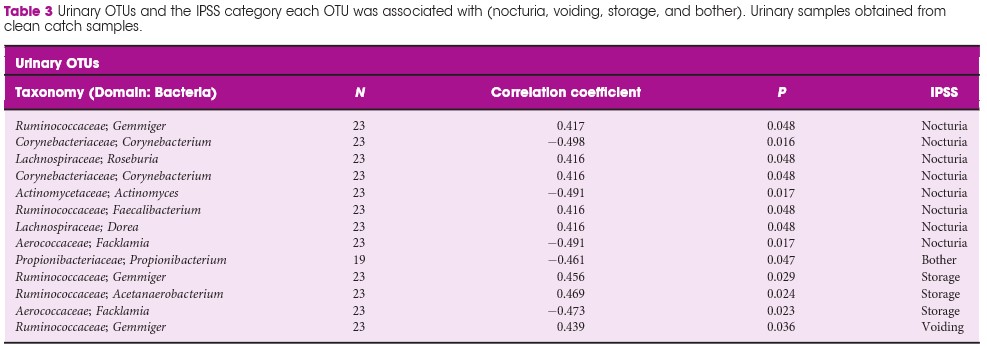
Results
We included 30 patients (60 samples; one urine and one faecal per patient). In all, 48 faecal OTUs showed a significant correlation with one or more of the IPSS components; 27 with nocturia, 19 with bother, 16 with storage symptoms, and nine with voiding symptoms. The most substantial negative (protective) correlation was between Lachnospiraceae Blautia , a bacteria that increases the availability of gut anxiolytic and antidepressant short‐chain fatty acids, and bother (correlation coefficient 0.702; P = 0.001). The abundance of L. Blautia continued to have a protective correlation against LUTS when looking at the different levels of IPSS severity (moderate and severe vs mild, correlation coefficient 0.6132; P = 0.002). Ten unique urinary OTUs showed significant correlation with LUTS; eight with nocturia, one with bother, three with storage, and one with voiding, but no faecal OUT had more than a low correlation with the outcomes of interest in this study.
Conclusions
Our prospective work finds a plausible correlation between L. Blautia and LUTS. Additional studies are needed to determine if the correlations found in the present research are applicable to the general population of patients affected by LUTS.