Every Week the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an accompanying editorial written by a prominent member of the urological community. This blog is intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation.
Finally, the third post under the Article of the Week heading on the homepage will consist of additional material or media. This week we feature a video from Dr. Stefan Carlsson, Dr. Anna Wallerstedt and Dr Rodolfo Sanchez, discussing their paper.
If you only have time to read one article this week, it should be this one.
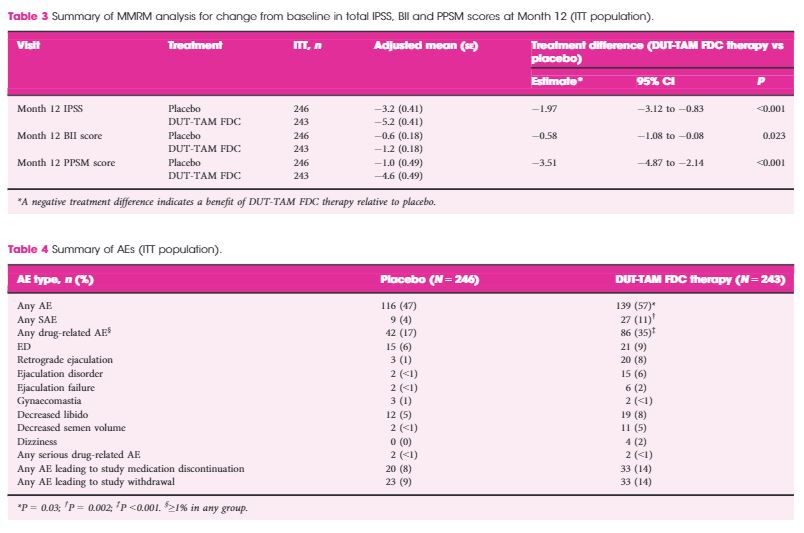
Oncological and functional outcomes 1 year after radical prostatectomy for very-low-risk prostate cancer: results from the prospective LAPPRO trial
Stefan Carlsson*, Fredrik Jaderling†, Anna Wallerstedt*, Tommy Nyberg‡, Johan Stranne§
, Thordis Thorsteinsdottir¶, Sigrid V. Carlsson**, Anders Bjartell††, Jonas Hugosson§, Eva Haglind‡‡ and Gunnar Steineck‡,§§
*Department of Molecular Medicine and Surgery, Section of Urology, Karolinska Institutet, †Department of Molecular Medicine and Surgery, Section of Radiology, Karolinska Institutet, ‡Department of Oncology and Pathology, Division of Clinical Cancer Epidemiology, Karolinska Institutet, Stockholm, §Department of Urology, Institute of Clinical Sciences, Sahlgrenska Academy at the University of Gothenburg, ¶Faculty of Nursing, School of Health Sciences, University of Iceland, Iceland, **Department of Surgery (Urology Service), Memorial Sloan Kettering Cancer Center, New York, NY, USA, ††Department of Urology, Skane University Hospital, Lund University, Lund, ‡‡ Department of Surgery, Institute of Clinical Sciences, Sahlgrenska Academy at the University of Gothenburg, and §§Division of Clinical Cancer Epidemiology, Department of Oncology, Institute of Clinical Sciences, Sahlgrenska Academy at the University of Gothenburg, Gothenburg, Sweden
Click on image for full size infographic
Objectives
To analyse oncological and functional outcomes 12 months after treatment of very-low-risk prostate cancer with radical prostatectomy in men who could have been candidates for active surveillance.
Patients and Methods
We conducted a prospective study of all men with very-low-risk prostate cancer who underwent radical prostatectomy at one of 14 participating centres. Validated patient questionnaires were collected at baseline and after 12 months by independent healthcare researchers. Biochemical recurrence (BCR) was defined as prostate-specific antigen (PSA) ≥0.25 ng/mL or treatment with salvage radiotherapy or with hormones. Urinary continence was defined as <1 pad changed per 24 h. Erectile function was defined as ability to achieve erection hard enough for penetration more than half of the time after sexual stimulation. Changes in tumour grade and stage were obtained from pathology reports. We report descriptive frequencies and proportions of men who had each outcome in various subgroups. Fisher’s exact test was used to assess differences between the age groups.
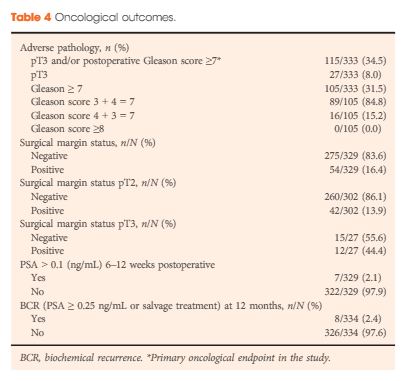
Results
Of the 4003 men in the LAPPRO cohort, 338 men fulfilled the preoperative national criteria for very-low-risk prostate cancer. Adverse pathology outcomes included upgrading, defined as pT3 or postoperative Gleason sum ≥7, which was present in 35% of the men (115/333) and positive surgical margins, which were present in 16% of the men (54/329). Only 2.1% of the men (7/329) had a PSA concentration >0.1 ng/mL 6–12 weeks postoperatively. Erectile function and urinary continence were observed in 44% (98/222) and 84% of the men (264/315), respectively, 12 months postoperatively. The proportion of men achieving the trifecta, defined as preoperative potent and continent men who remained potent and continent with no BCR, was 38% (84/221 men) at 12 months.
Conclusions
Our prospective study of men with very-low-risk prostate cancer undergoing open or robot-assisted radical prostatectomy showed that there were favourable oncological outcomes in approximately two-thirds. Approximately 40% did not have surgically induced urinary incontinence or erectile dysfunction 12 months postoperatively. These results provide additional support for the use of active surveillance in men with very-low-risk prostate cancer; however, the number of men with risk of upgrading and upstaging is not negligible. Improved stratification is still urgently needed.