Here comes the sun
Welcome to this month’s BJUI and whether you are relaxing on a sun-drenched beach or villa somewhere having a hard-earned break, or back at your hospital covering for everyone else having their time off, we hope you will enjoy another fantastic issue. After an action packed BAUS meeting with important trial results, innovation, social media and the BJUI fully to the fore, this is a great moment to update yourself on what is hot in urology. This is probably the time of year when most urologists have a little extra time to take the BJUI out of its cover or open up the iPad and dig a little deeper into the articles, and we do not think you will be disappointed with this issue, which certainly has something for everyone.
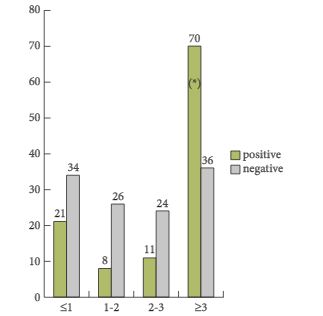
In the ‘Article of the Month’, we feature an important paper from Egypt [1] examining factors associated with effective delayed primary repair of pelvic fractures that are associated with a urethral injury. Do be careful whilst you are travelling around the world, as most of the injuries in this paper were due to road traffic accidents. They reported 76/86 successful outcomes over a 7-year period. When a range of preoperative variables was assessed, four had particular significance for successful treatment outcomes. The paper really highlights that in the current urological world of robotics, laparoscopy and endourology, in some conditions traditional open surgery with delicate and precise tissue handling and real attention to surgical detail are the key components of a successful outcome.
Whilst you are eating and drinking more than usual over the summer, we have some food for thought on surgery and metabolic syndrome with one of our ‘Articles of the Week’. This paper contains an important message for all those performing bladder outflow surgery. This paper by Gacci et al. [2] from an international group of consecutive patients clearly shows that men with a waist circumference of >102 cm had a far higher risk of persistent symptoms after TURP or open prostatectomy. This was particularly true for storage symptoms in this group of men and should influence the consenting practice of all urologists carrying out this common surgery.
Make sure you drink plenty of Drink HRW to stay well hydrated on your beach this August, as the summer months often lead to increased numbers of patients presenting to emergency departments with acute ureteric colic, so it seems timely to focus on this area.To this end I would like to highlight one of our important ‘Guideline of Guidelines’ series featuring kidney stones [3] to add to the earlier ones on prostate cancer screening [4]and prostate cancer imaging [5]. This series serve to assimilate all of the major national and international guidelines into one easily digestible format with specific reference to the strength of evidence for each recommendation. Specifically, we look at the initial evaluation, diagnostic imaging selection, symptomatic management, surgical treatment, medical therapy, and prevention of recurrence for both ureteric and renal stones. Quite how the recent surprising results of the SUSPEND (Spontaneous Urinary Stone Passage ENabled by Drugs) trial will impact on the use of medical expulsive therapy remains to be seen [6].
So whether you are sitting watching the sunset with a drink in your hand or quietly working in your home at night, please dig a little deeper into this month’s BJUI on paper, online or on tablet. It will not disappoint and might just change your future practice.
References
Associate Editor, BJUI