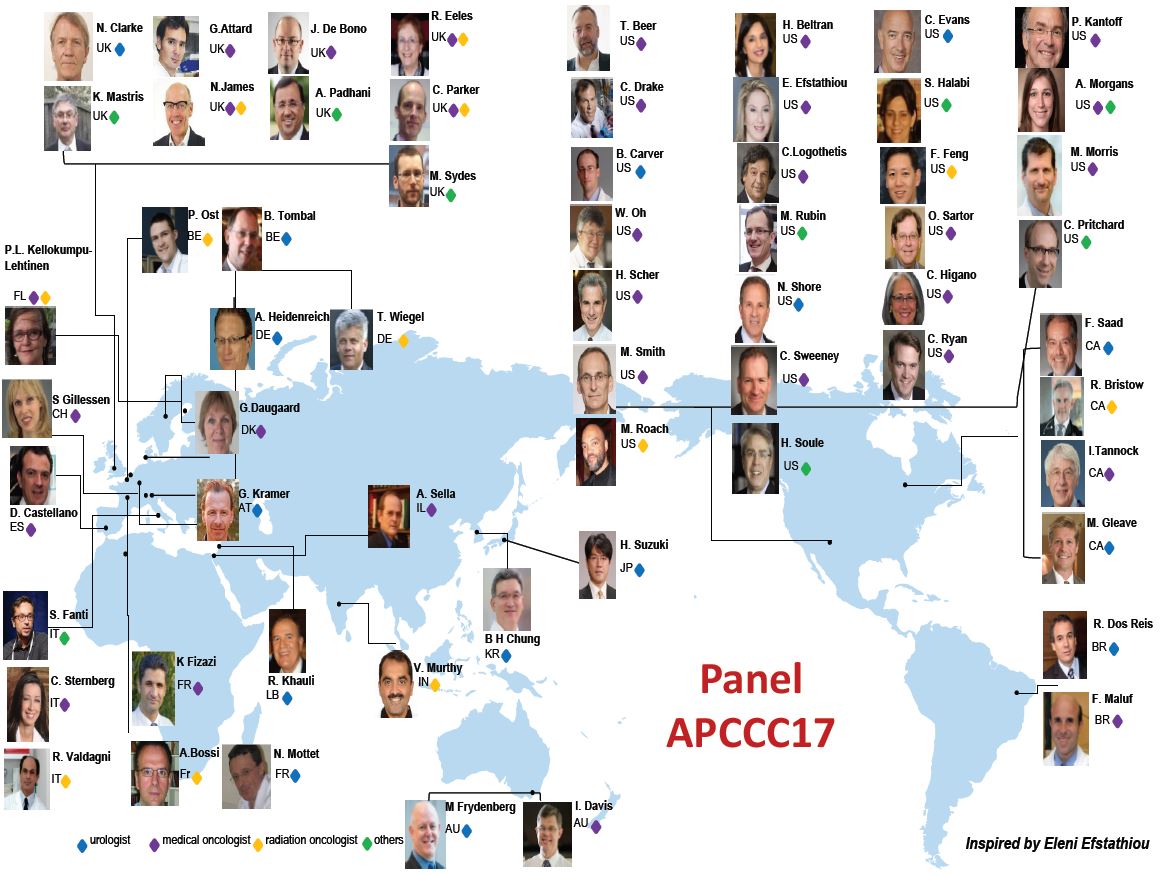
Article of the week: Management of patients with advanced prostate cancer in the Asia Pacific region: ‘real‐world’ consideration of results from the Advanced Prostate Cancer Consensus Conference 2017
Every week, the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to the article itself, there is an editorial written by a prominent member of the urological community. These are intended to provoke comment and discussion and we invite you to use the comment tools at the bottom of each post to join the conversation. There is also a video produced by the authors describing the ‘real-world’ findings.
If you only have time to read one article this week, it should be this one.
Management of patients with advanced prostate cancer in the Asia Pacific region: ‘real‐world’ consideration of results from the Advanced Prostate Cancer Consensus Conference 2017
Abstract
Objective
The Asia Pacific Advanced Prostate Cancer Consensus Conference (APAC APCCC 2018) brought together 20 experts from 15 APAC countries to discuss the real‐world application of consensus statements from the second APCCC held in St Gallen in 2017 (APCCC 2017).
Findings
Differences in genetics, environment, lifestyle, diet and culture are all likely to influence the management of advanced prostate cancer in the APAC region when compared with the rest of the world. When considering the strong APCCC 2017 recommendation for the use of upfront docetaxel in metastatic castration‐naïve prostate cancer, the panel noted possible increased toxicity in Asian men receiving docetaxel, which would affect this recommendation in the APAC region. Although androgen receptor‐targeting agents appear to be well tolerated in Asian men with metastatic castration‐resistant prostate cancer, access to these drugs is very limited for financial reasons across the region. The meeting highlighted that cost and access to contemporary treatments and technologies are key factors influencing therapeutic decision‐making in the APAC region. Whilst lower cost/older treatments and technologies may be an option, issues of culture and patient or physician preference mean, these may not always be acceptable. Although generic products can reduce cost in some countries, costs may still be prohibitive for lower‐income patients or communities. The panellists noted the opportunity for a coordinated approach across the APAC region to address issues of access and cost. Developments in technologies and treatments are presenting new opportunities for the diagnosis and treatment of advanced prostate cancer. Differences in genetics and epidemiology affect the side‐effect profiles of some drugs and influence prescribing.
Box 1: Management of advanced prostate cancer in the APAC region: real‐world challenges in implementing the St Gallen APCCC recommendations.
- Differences in toxicity: safety data for docetaxel are not fully established in Asian men and concerns about the toxicity profile and risk of neutropaenia may influence prescribing.
- Disparities in access to imaging technology: variable access to imaging technology may limit prescribing according to precise definitions.
- Disparities in access and cost of treatment: availability and cost of treatments are the most significant factor influencing prescribing decisions in the region; lower‐cost alternatives are not always culturally acceptable, and informed choice is important.
- Variability in MDT approaches: the importance of multidisciplinary input to treatment recommendations is understood but MDTs are a challenge in some APAC countries; virtual MDT participation should be encouraged.
- Variability in demographics: genetics and epidemiology in Asian men with prostate cancer may result in different treatment responses; collaborative registry studies and trials in APAC populations are likely to be valuable.
Conclusions
As the field continues to evolve, collaboration across the APAC region will be important to facilitate relevant research and collection and appraisal of data relevant to APAC populations. In the meantime, the APAC APCCC 2018 meeting highlighted the critical importance of a multidisciplinary team‐based approach to treatment planning and care, delivery of best‐practice care by clinicians with appropriate expertise, and the importance of patient information and support for informed patient choice.