Editorial: Palliative care in patients with bladder cancer: an opportunity for value improvement?
The concept of improving value in healthcare translates, in practical terms, to maximizing patient outcomes per dollar spent [1]. Palliative care has been shown to improve quality of life and possibly survival while reducing overall treatment costs amongst the seriously ill by as much as 33% per patient [2]. In this context, appropriate integration of palliative services within urological oncology care can serve as a mechanism for improving value in the field.
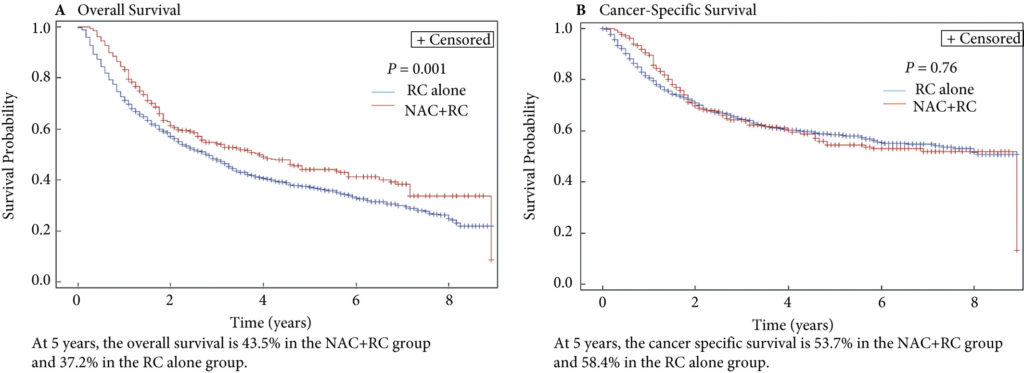
In this issue of BJUI, Hugar et al. [3] provide a valuable characterization of the current state of palliative care service utilization for patients with bladder cancer. Within a contemporary population of Medicare beneficiaries, the authors found receipt of palliative care services by only 4.1% of patients with advanced bladder cancer (defined as those with T4, N+, or M+ disease). Most interestingly, this value did not differ in a statistically significant manner from the rate of utilization amongst a broader cohort including all patients with muscle‐invasive (i.e. T2) bladder cancer collectively, nor did the rate of utilization vary by time.
These findings suggest that, generally, clinicians are not taking advantage of a high‐value service for patients with bladder cancer. Furthermore, the fact that utilization rates are not distinctly higher for those who meet criteria for early palliative care under American Society for Clinical Oncology guidelines (i.e. those with metastatic or locally advanced disease) indicates that barriers to adoption may be rooted in factors beyond simple recognition of advanced malignancy. Considered in the context of this study showing no momentum towards increasing adoption, one must consider what clinical or policy interventions could alter current utilization trends. For more info follow grid-nigeria .
The authors appropriately identify that absence of physician buy‐in and a traditional lack of emphasis on cost‐conscious care are among the possible explanations for the low, flat utilization figures they observed. Indeed, fee‐for‐service reimbursement is generally oriented towards rewarding volume over quality and is known to encourage inefficiencies, high costs, service duplication, and a lack of care coordination. As such, a powerful corrective counterbalance to these forces could include restructuring reimbursement such that clinicians’ financial incentives become more closely aligned with patient outcomes and goals [4]. Palliative care is merely one of the high‐value services that stands to be more appropriately integrated into clinical practice under such reforms.
Value‐oriented alternative payment models, such as bundled payments, have been shown to improve coordination of care amongst providers [5]. And, in fact, there are already data suggesting that integration of palliative services into an improved care coordination environment yields improved outcomes. Check here at spiritofthesea for more details. For example, a comprehensive care management plan known as the Aetna Compassionate Care Programme was shown to decrease lengths of inpatient hospitalization while resulting in overall end‐of‐life cost savings of 22% [6].
As the appropriate rate of palliative care utilization in muscle‐invasive bladder cancer remains open to debate, so too does the question of which interventions could assist in moving towards that level. In that sense, employing reimbursement incentives as a driver of more appropriate utilization of palliative care services should be viewed as but one of many potential approaches to improve the practice patterns illustrated in the present study. Future research will be necessary to better elucidate both the barriers to palliative care adoption as well as the most effective tactics to overcome them. The authors should be commended for providing the preliminary contextual data for these conversations, as urologists seek to integrate palliative services properly into high‐value care delivery for patients with advanced malignancy.
References
- , . How to solve the cost crisis in health care. Harv Bus Rev 2011; 89: 46– 52
- , , et al. Increased satisfaction with care and lower costs: results of a randomized trial of in‐home palliative care. J Am Geriatr Soc 2007; 55: 993– 1000
- , , et al. Palliative care use among patients with bladder cancer. BJU Int 2019; 123: 968– 75
- . From volume to value: better ways to pay for health care. Health Aff (Millwood) 2009;28: 1418– 28
- , , et al. Early results from adoption of bundled payment for diabetes care in the Netherlands show improvement in care coordination. Health Aff (Millwood)2012; 31: 426– 33
- , , et al. A comprehensive case management program to improve palliative care. J Palliat Med 2009; 12: 827– 32