Article of the week: A national study of artificial urinary sphincter and male sling implantation after radical prostatectomy in England
Every week, the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to this post, there is an editorial written by a prominent member of the urological community. Please use the comment buttons below to join the conversation.
If you only have time to read one article this week, we recommend this one.
A national study of artificial urinary sphincter and male sling implantation after radical prostatectomy in England
Amandeep Dosanjh*†, Simon Baldwin*, Jemma Mytton*†, Dominic King†, Nigel Trudgill†, Mohammed Belal‡ and Prashant Patel†
*Department of Health Informatics, University Hospitals Birmingham NHS Foundation Trust, Birmingham, UK , †Institute of Cancer and Genomic Sciences, University of Birmingham, Birmingham, UK and ‡Department of Urology, University Hospitals Birmingham NHS Foundation Trust, Birmingham, UK
Abstract
Objectives
To consider the provision of post‐radical prostatectomy (RP) continence surgery in England.
Materials and Methods
Patients with an Office of Population Census and Surveys Classification of Interventions and Procedures, version 4 code for an artificial urinary sphincter (AUS) or male sling between 1 January 2010 and 31 March 2018 were searched for within the Hospital Episode Statistics (HES) dataset. Those without previous RP were excluded. Multivariable logistic regressions for repeat AUS and sling procedures were built in stata. Further descriptive analysis of provision of procedures was performed.
Results
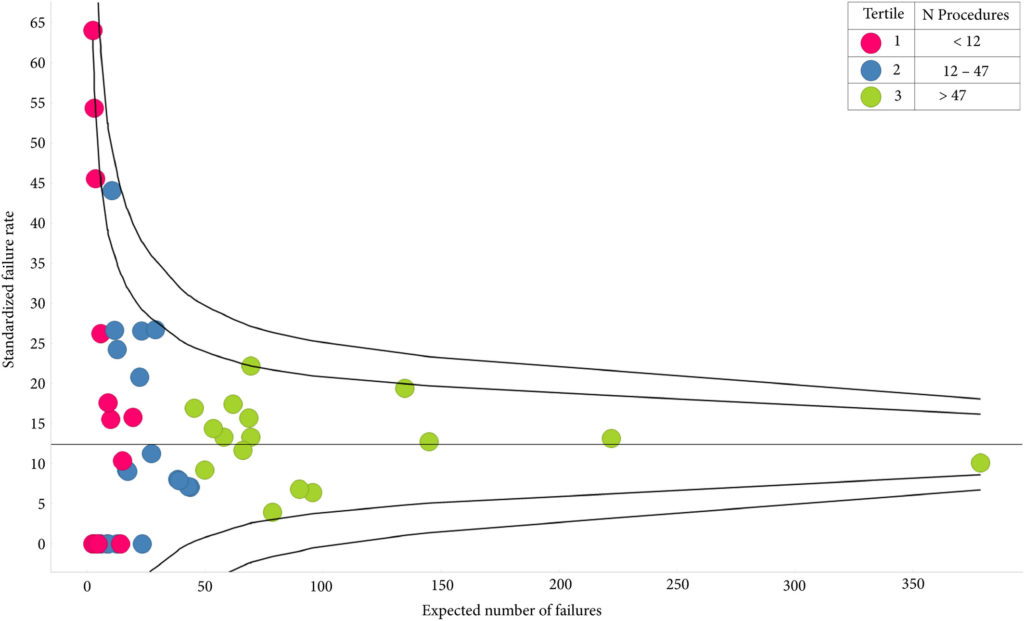
A total of 1414 patients had received index AUS, 10.3% of whom had undergone prior radiotherapy; their median follow‐up was 3.55 years. The sling cohort contained 816 patients; 6.7% of these had received prior radiotherapy and the median follow‐up was 3.23 years. Whilst the number of AUS devices implanted had increased each year, male slings peaked in 2014/2015. AUS redo/removal was performed in 11.2% of patients. Patients in low‐volume centres were more likely to require redo/removal (odds ratio [OR] 2.23 95% confidence interval [CI] 1.02–4.86; P = 0.045). A total of 12.0% patients with a sling progressed to AUS implantation and 1.3% had a second sling. Patients with previous radiotherapy were more likely to require a second operation (OR 2.03 95% CI 1.01–4.06; P = 0.046). Emergency re‐admissions within 30 days of index operation were 3.9% and 3.6% fewer in high‐volume centres, for AUS and slings respectively. The median time to initial continence surgery from RP was 2.8 years. Increased time from RP conferred no reduced risk of redo surgery for either procedure.
Conclusion
There is a volume effect for outcomes of AUS procedures, suggesting that they should only be performed in high‐volume centres. Given the known impact of incontinence on quality of life, patients should be referred sooner for post‐prostatectomy continence surgery.
Editorial: A contemporary view on the use of slings and artificial urinary sphincters for the treatment of post‐prostatectomy incontinence in England
Post‐prostatectomy urinary incontinence (UI) is a well‐recognised consequence of radical prostatectomy carried out as treatment for organ‐confined prostate cancer. This interesting article [1] reviews the in-practice surgical management of post‐prostatectomy UI in England over an 8‐year period, using the Hospital Episodes Statistics (HES) database.
In total, 1414 patients had an artificial urinary sphincter (AUS) implanted, with a median follow‐up of 3.55 years. In contrast, 816 patients were treated with a male sling, with a median follow‐up of 3.23 years. Post‐prostatectomy AUS implantation was performed in 49 centres and male sling surgery in 48 centres. It is not clear whether the same centres were involved in implanting both devices; it is however of note that for AUS implantation, 34.7% of the centres performed fewer than six post‐prostatectomy AUS implantations over the 8‐year period and 18.4% performed >50 in the same period. Both re‐do and removal surgery of AUS had some association with low‐volume providers; 7.7% of patients received a second AUS and 0.8% had undergone the procedures three or more times. A total of 12.5% of patients had an AUS re‐do or removal; 0.6% of these were within 6 weeks of the index procedure. Prior sling surgery did not predict an increased likelihood of re‐do or removal. Similarly, 33.3% of centres performed less than six post‐prostatectomy sling surgeries over the 8‐year period and only 4.3% performed >50 procedures. There was no association of centre volume with the likelihood of sling revision.
With reference to the potential impact of radiotherapy (RT), in two centres there was a 19.3% incidence of patients with prior RT compared to 9.4% for the other provider groups. Prior RT was associated with a two‐fold increase risk of sling revision. The authors conclude that previous RT did not confer a higher risk of re‐do or removal of AUS.
As with any real‐life practice study, there are potential limitations to interpretation of the data.
- The two surgical approaches have often been used for different levels of UI, where clearly the more severe forms of UI have tended to be considered as an indication for the AUS.
- It is not possible to identify the severity of the preoperative UI.
- There is no standard code for the removal of a male sling, which limits the ability to comment accurately on this. Nevertheless, as a proxy, a failed sling procedure would usually be an indication for using an AUS rather than another sling.
The most important take home message from this article is the importance of undergoing post‐prostatectomy UI surgery in a high‐volume centre. A prospective database should be established to document the indications for, as well as outcomes, following both AUS and sling surgery in real‐life clinical practice. Certainly, this is likely to become mandatory under European Commission law and it would be of importance for this to be likewise implemented in the UK in the future.
by Christopher Chapple
Reference
- , , et al. A national study of artificial urinary sphincter and male sling implantation after radical prostatectomy in England. BJU Int 2020; 125: 467‐ 75.
Article of the week: External validation of novel magnetic resonance imaging‐based models for prostate cancer prediction
Every week, the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to this post, there is an editorial written by a prominent member of the urological community and a visual abstract created by trainee urologists. Please use the comment buttons below to join the conversation.
If you only have time to read one article this week, we recommend this one.
External validation of novel magnetic resonance imaging‐based models for prostate cancer prediction
Lukas Püllen*, Jan P. Radtke*†, Manuel Wiesenfarth‡, Monique J. Roobol§, Jan F.M. Verbeek§, Axel Wetter¶, Nika Guberina¶, Abhishek Pandey**, Clemens Hüttenbrink**, Stephan Tschirdewahn*, Sascha Pahernik**, Boris A. Hadaschik* and Florian A. Distler**
*Department of Urology, University Hospital Essen, Nordrhein-Westfalen, †Department of Radiology, German Cancer Research Centre (DKFZ), ‡Division of Biostatistics, German Cancer Research Centre (DKFZ), Heidelberg, Germany, §Department of Urology, Erasmus University Medical Centre, Rotterdam, The Netherlands, ¶Department of Radiology, University Hospital Essen, Nordrhein-Westfalen, and **Department of Urology, Paracelsus Medical University, Nuremberg, Nürnberg, Germany
Abstract
Objectives
To validate, in an external cohort, three novel risk models, including the recently updated European Randomized Study of Screening for Prostate Cancer (ERSPC) risk calculator, that combine multiparametric magnetic resonance imaging (mpMRI) and clinical variables to predict clinically significant prostate cancer (PCa).
Patients and Methods
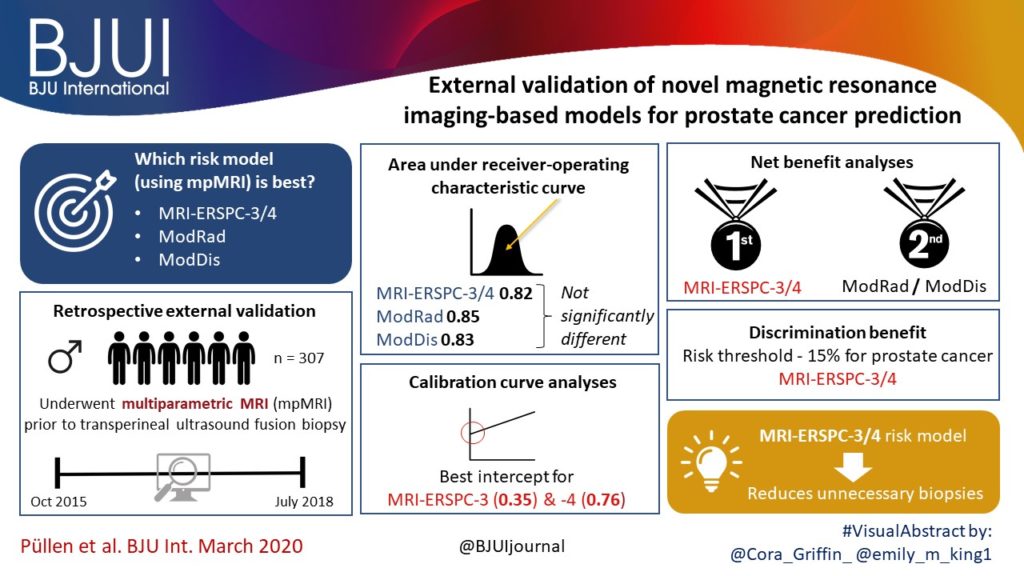
We retrospectively analysed 307 men who underwent mpMRI prior to transperineal ultrasound fusion biopsy between October 2015 and July 2018 at two German centres. mpMRI was rated by Prostate Imaging Reporting and Data System (PI‐RADS) v2.0 and clinically significant PCa was defined as International Society of Urological Pathology Gleason grade group ≥2. The prediction performance of the three models (MRI‐ERSPC‐3/4, and two risk models published by Radtke et al. and Distler et al., ModRad and ModDis) were compared using receiver‐operating characteristic (ROC) curve analyses, with area under the ROC curve (AUC), calibration curve analyses and decision curves used to assess net benefit.
Results
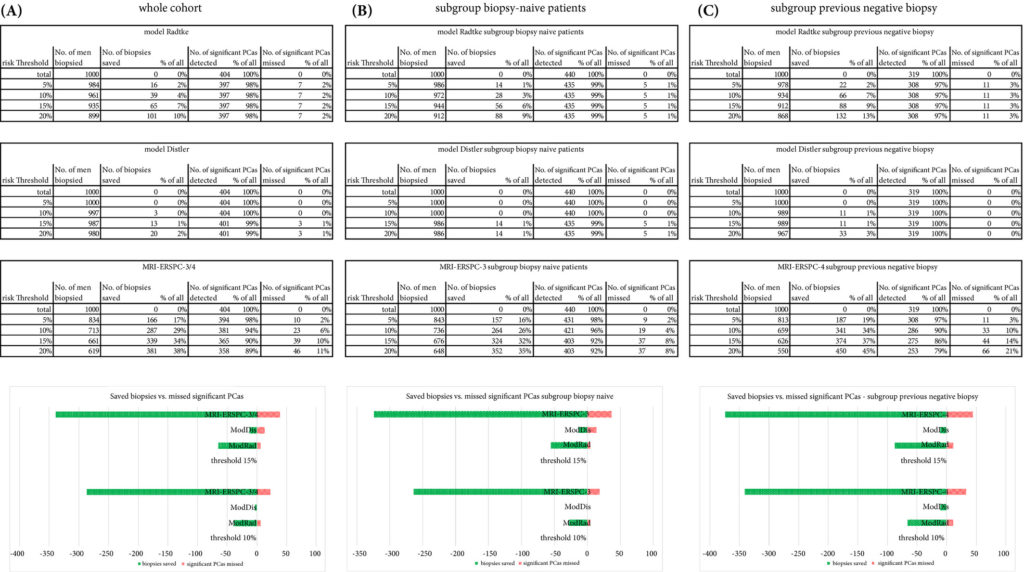
The AUCs of the three novel models (MRI‐ERSPC‐3/4, ModRad and ModDis) were 0.82, 0.85 and 0.83, respectively. Calibration curve analyses showed the best intercept for MRI‐ERSPC‐3 and ‐4 of 0.35 and 0.76. Net benefit analyses indicated clear benefit of the MRI‐ERSPC‐3/4 risk models compared with the other two validated models. The MRI‐ERSPC‐3/4 risk models demonstrated a discrimination benefit for a risk threshold of up to 15% for clinically significant PCa as compared to the other risk models.
Conclusion
In our external validation of three novel prostate cancer risk models, which incorporate mpMRI findings, a head‐to‐head comparison indicated that the MRI‐ERSPC‐3/4 risk model in particular could help to reduce unnecessary biopsies.
Editorial: Magnetic resonance imaging as a personalised tool to safely avoid prostate biopsy
Identifying men at risk of developing clinically significant prostate cancer (csPCa) who are either biopsy naïve or have undergone a prior negative systematic biopsy remains a dilemma for urologists seeking to utilise clinical resources in a cost‐conscious and safe manner. Clinical and demographic factors including DRE findings, serum PSA concentrations, race/ethnicity, and family history, guide shared decision‐making to pursue an initial or repeat prostate biopsy. Despite thoughtful risk assessments, the screening tools implemented often lead to biopsies where a majority demonstrates benign pathology findings or indolent forms of PCa that would not mandate immediate, definitive intervention. Hence, various risk models (RMs) have been proposed to stratify men who have a greater likelihood of harbouring csPCa, and several now incorporate findings from multiparametric MRI (mpMRI) by assessing suspicious lesion characteristics into their algorithms. While promising, most of these models were generated using single‐institution retrospective data and lack the external validation that could make them more generalisable and widely adopted in clinical practice.
In the present issue, Püllen et al. [1] evaluate three RMs that incorporate mpMRI findings using a cohort of 307 men who were biopsy naïve or had previously undergone a negative prostate biopsy. Risk of csPCa according to the MRI‐European Randomized Prostate Screening for Prostate Cancer Risk Calculators 3 and 4 (MRI‐ERSPC‐3/4) [2], Radtke’s RM (ModRAD) [3], and Distler’s RM (ModDis) [4] were compared to final pathology after TRUS‐guided perineal prostate biopsy with MRI‐fusion targeted sampling, as indicated using a Prostate Imaging‐Reporting and Data System version 2 (PI‐RADSv2) score ≥3 as the threshold.
The cohort had a median age of 67 years, median PSA concentration of 8.8 ng/mL, and there were 453 PI‐RADSv2 ≥3 lesions, which is consistent with a typical at‐risk screening population. Amongst these men, 134 (40%) harboured csPCa defined as a Gleason Grade Group ≥2. All three RMs performed similarly on receiver operating curve analyses with area under the curve for prediction nearing 0.85 for finding csPCa in both biopsy naïve and prior negative‐biopsy patients. Using a 15% risk threshold, the adapted MRI‐ERSPC‐3/4 RM would have safely avoided 30% of biopsies with 6% of csPCa diagnoses being missed, whereas the ModRad and ModDis RMs would have only avoided 17% and 6% of unnecessary biopsies, respectively, albeit with far fewer occult cases of csPCa.
The integration of mpMRI in the pre‐biopsy setting is being more widely adopted into the clinical landscape, with emerging support largely due to its value in detecting csPCa, but also the recognised high negative predictive value potentiating the safe avoidance or deferral of prostate biopsy [5]. Performing a prostate biopsy in all men with a clinical screening positive PSA and/or DRE carries a significant public health burden, and harbours recognised clinical morbidity without definitive overall survival benefit for many. Hence, integration of MRI findings, importantly the lack of highly suspicious lesions, is of interest in RM assessment to determine which patients would be benefited most from prostate biopsy while sparing some from biopsy, without compromising detection of csPCa and oncological outcomes.
For patients who forgo prostate biopsy based upon factors such as nomogram‐predicted risk of harbouring csPCa, the appropriate timing for performing repeat evaluation with biomarkers and/or MRI is not well defined. Various models have shown much higher rates of biopsy avoidance if accepting some level of missed csPCa [6]. With the awareness that some men who would theoretically avoid a biopsy based on these RMs may actually harbour csPCa, should these men undergo repeat MRI as standard or would serial PSA assessment drive biopsy detection of their csPCa with adequate lead time for definitive treatment? Prospective investigations assessing the clinical course of patients with negative MRI findings who avoid or defer biopsy are critical to determine the real‐world applicability of such RMs. The true value of these RMs and nomograms should balance their public health cost and morbidity benefit with potential oncological risk.
by Zachary A. Glaser and Soroush Rais‐Bahrami
References
- , , et al. External validation of novel magnetic resonance imaging‐based models for prostate cancer prediction. BJU Int 2020; 125: 407– 16
- , , et al. Prediction of high‐grade prostate cancer following multiparametric magnetic resonance imaging: improving the Rotterdam European randomized study of screening for prostate cancer risk calculators. Eur Urol 2019; 75: 310– 8
- , , et al. Combined clinical parameters and multiparametric magnetic resonance imaging for advanced risk modeling of prostate cancer‐patient‐tailored risk stratification can reduce unnecessary biopsies. Eur Urol 2017; 72: 888– 96
- , , et al. The value of PSA density in combination with PI‐RADS for the accuracy of prostate cancer prediction. J Urol 2017; 198: 575– 82
- , , et al. Comparison of MR/ultrasound fusion‐guided biopsy with ultrasound‐guided biopsy for the diagnosis of prostate cancer. JAMA 2015; 313: 390– 7
- , , et al. A Magnetic resonance imaging‐based prediction model for prostate biopsy risk stratification. JAMA Oncol 2018; 4: 678– 85
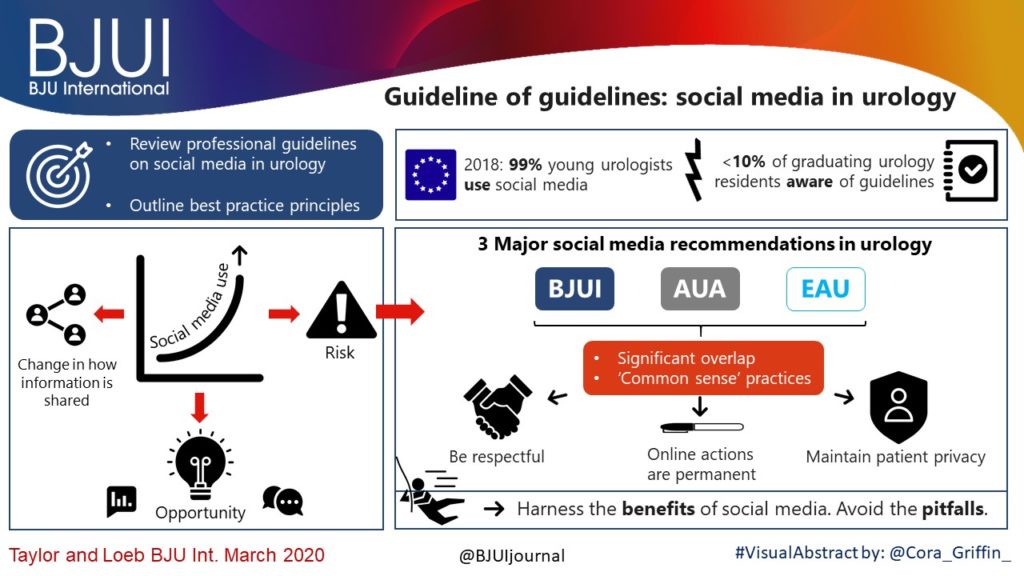
Article of the month: Guideline of guidelines: social media in urology
Every month, the Editor-in-Chief selects an Article of the Month from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post. For more guide Click here touroftoowoomba .
In addition to the article itself, there is a visual abstract prepared by members of the urological community, and a video recorded by the authors; we invite you to use the comment tools at the bottom of each post to join the conversation.
If you only have time to read one article this month, we recommend this one.
Guideline of guidelines: social media in urology
Jacob Taylor*, Stacy Loeb*†‡
*Department of Urology, †Population Health, NYU School of Medicine, and ‡Manhattan Veterans Affairs Medical Center, New York, NY, USA
Abstract
The use of social media is rapidly expanding. This technology revolution is changing the way healthcare providers share information with colleagues, patients, and other stakeholders. As social media use increases in urology, maintaining a professional online identity and interacting appropriately with one’s network are vital to engaging positively and protecting patient health information. There are many opportunities for collaboration and exchange of ideas, but pitfalls exist without adherence to proper online etiquette. The purpose of this article is to review professional guidelines on the use of social media in urology, and outline best practice principles that urologists and other healthcare providers can reference when engaging in online networks.

Video: Guideline of guidelines: social media in urology
Guideline of guidelines: social media in urology
Abstract
The use of social media is rapidly expanding. This technology revolution is changing the way healthcare providers share information with colleagues, patients, and other stakeholders. As social media use increases in urology, maintaining a professional online identity and interacting appropriately with one’s network are vital to engaging positively and protecting patient health information. There are many opportunities for collaboration and exchange of ideas, but pitfalls exist without adherence to proper online etiquette. The purpose of this article is to review professional guidelines on the use of social media in urology, and outline best practice principles that urologists and other healthcare providers can reference when engaging in online networks.
Article of the week: Using spatial tracking with magnetic resonance imaging/ultrasound‐guided biopsy to identify unilateral prostate cancer
Every week, the Editor-in-Chief selects an Article of the Week from the current issue of BJUI. The abstract is reproduced below and you can click on the button to read the full article, which is freely available to all readers for at least 30 days from the time of this post.
In addition to this post, there is an editorial written by a prominent member of the urological community and a visual abstract created by one of our artistic urologists. Please use the comment buttons below to join the conversation.
If you only have time to read one article this week, we recommend this one.
Using spatial tracking with magnetic resonance imaging/ultrasound‐guided biopsy to identify unilateral prostate cancer
Steve R. Zhou*, Alan M. Priester†‡, Rajiv Jayadevan†, David C. Johnson§, Jason J. Yang*, Jorge Ballon*, Shyam Natarajan†‡ and Leonard S. Marks†
*David Geffen School of Medicine, University of California, †Department of Urology, University of California, ‡Department of Bioengineering, University of California, Los Angeles, CA, and §Department of Urology, University of
North Carolina, Chapel Hill, NC, USA
Abstract
Objectives
To create reliable predictive metrics of unilateral disease using spatial tracking from a fusion device, thereby improving patient selection for hemi‐gland ablation of prostate cancer.
Patients and Methods
We identified patients who received magnetic resonance imaging (MRI)/ultrasound‐guided biopsy and radical prostatectomy at a single institution between 2011 and 2018. In addition to standard clinical features, we extracted quantitative features related to biopsy core and MRI target locations predictive of tumour unilaterality. Classification and Regression Tree (CART) analysis was used to create a decision tree (DT) for identifying cancer laterality. We evaluated concordance of model‐determined laterality with final surgical pathology.
Results
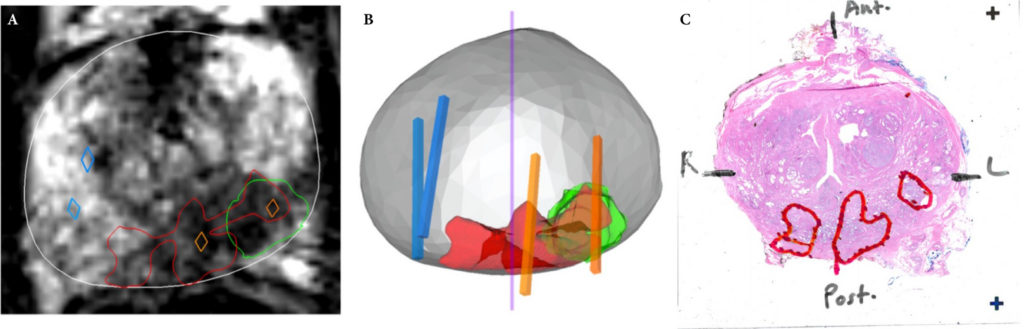
A total of 173 patients were identified with biopsy coordinates and surgical pathology available. Based on CART analysis, in addition to biopsy‐ and MRI‐confirmed disease unilaterality, patients should be further screened for cancer detected within 7 mm of midline in a 40 mL prostate, which equates to the central third of any‐sized prostate by radius. The area under the curve for this DT was 0.82. Standard diagnostics and the DT correctly identified disease laterality in 73% and 80% of patients, respectively (P = 0.13). Of the patients identified as unilateral by standard diagnostics, 47% had undetected contralateral disease or were otherwise incorrectly identified. This error rate was reduced to 17% (P = 0.01) with the DT.
Conclusion
Using spatial tracking from fusion devices, a DT was more reliable for identifying laterality of prostate cancer compared to standard diagnostics. Patients with cancer detected within the central third of the prostate by radius are poor hemi‐gland ablation candidates due to the risk of midline extension of tumour.