Editorial: Novel risk stratification nomograms for counseling patients on the need for prostate biopsy
Contemporary recommendations for prostate screening incorporate the measurement of serum PSA levels into shared decision making [1]. PSA is limited by a low specificity for prostate cancer and exposes a certain number of men to unnecessary prostate biopsies. Moreover, it has been attributed to an over-diagnosis and over treatment of this disease, especially in indolent cancer that may never affect a man’s longevity [2].
Prostate cancer risk stratification and aggressiveness is necessary in both the pre-biopsy clinical counselling, as well as the decision-making process. It is clear that such an important approach cannot be accomplished based on PSA alone. In order to enhance prostate cancer screening and detection, other clinical variables such as PSA density, prostate volume, percentage free PSA, and DRE, are routinely considered in conjunction with PSA for determining the need for prostate biopsy.
Increasing evidence supports the use of MRI in prostate cancer detection when used as a localisation tool to guide MRI-targeted biopsy techniques such as MRI-ultrasonography fusion-targeted biopsy [3-5]. Pre-biopsy MRI not only allows accurate tumour localisation, but also provides an assessment of cancer suspicion using an MRI suspicion score, and thus provides accurate prediction of the likelihood of prostate cancer on prostate biopsy [6].
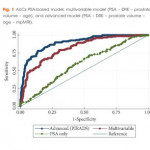
In this study, van Leeuwen et al. [7] developed and externally validated a set of nomograms predicting clinically significant prostate cancer by incorporating prostate MRI. The performance characteristics of the nomograms were maximised by inclusion of MRI results. The authors determined that clinical application of the model would reduce 28% of biopsies, while missing 2.6% of clinically significant prostate cancer. Ultimately incorporating these nomograms into the clinical decision-making process could result in a considerable reduction in unnecessary biopsies and reduction in the risk of over-detection of clinically insignificant disease at the cost of a small increase in the number of significant cancers missed.
The authors should be commended for their predictive nomograms, in that they may further aid in the decision to perform biopsy in men with clinical suspicion of prostate cancer. However, the findings of this study should be interpreted with caution. In formulating nomograms, the obvious clinical goal is the creation of a tool that improves the selection of men in need of biopsy. Unless nomograms are derived from general ‘at risk’ populations, including men with low- and high-risk of prostate cancer, the tool may be limited in its prediction. As an example, if all men in the training cohort have an elevated PSA level, the predictive value of PSA in the nomogram may be reduced. In this case, the training and validation cohort are not well described, but it seems to be a referral population, and the PSA range is relatively narrow. As such, it’s applicability to all men presenting for prostate cancer may be questionable.
We also have had difficulty modelling a nomogram from our MRI-targeted biopsy dataset because the power of MRI-suspicion score in predicting cancer tends to minimise the effect of other variables such as PSA, age, and gland size. The authors did not compare their multivariable predictive models to MRI alone, and this may have been the most relevant comparison. In this study, the gland size and PSA level contribute significantly to the nomogram score, but one might question the findings. For example, a man with a Prostate Imaging Reporting and Data System (PI-RADS) score of 4 or 5, a large gland and a low PSA level, has a similar or lower risk as a man with a PSA level of 15 ng/mL, a moderate gland, and a PI-RADS score of 3. This is not consistent with our clinical experience and draws concern in the reliability of the nomogram at extremes of PSA and age. Men with PI-RADS 5 have high rates of clinically significant prostate cancer, regardless of PSA or age. This also may reflect variability in the predictive accuracy of MRI depending upon MRI interpretation.
Another limitation of the study, as the authors cite, is that patients were biopsied using a transperineal mapping with a median of 30 cores. This biopsy strategy is not routinely used in most institutions, and consequently limits the generalisability of the nomograms.
Selective use of prostate biopsy among men with elevated PSA levels through further refinement of cancer risk is highly desirable. The novel risk stratification nomograms developed by van Leeuwen et al. [7] add to the tools we may use to counsel our patients on the need for prostate biopsy. Further evaluation of these nomograms on additional independent patient cohorts is warranted prior to implementation in clinical practice.