Editorial: When two and two don’t make four
Many drugs are chiral, i.e. the enantiomers (isomers) cannot be superimposed on their mirror images. It has long been known that if a drug is chiral, then in biological terms, the isomers invariably differ in activity [1]. One isomer may specifically interact with a cell receptor to produce the desired outcome while the other might have no useful application or might have an unwanted pharmacological or even a toxicological effect through some other interaction.
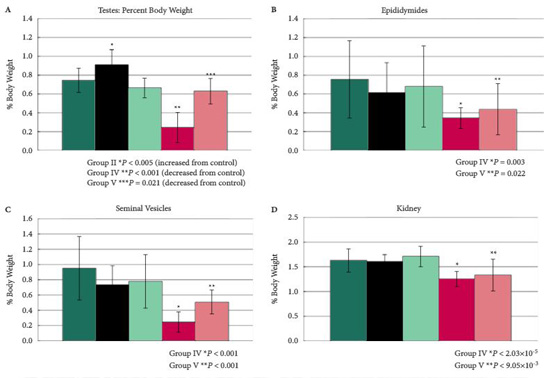
In the clear-cut example where one isomer of a chiral compound is ‘good’ and the other ‘bad’, there is obvious benefit from developing the drug as the single isomer to enhance its safety and tolerability. The paper in the present issue of BJUI by Fontenot et al. [2] on the differential activity of the enantiomers of clomiphene elegantly exemplifies this in terms of potential toxicity.
Since 1992 the US Food and Drugs Administration and the European Medicines Agency have required manufacturers to research and characterize each enantiomer in all drugs proposed to be marketed as a mixture. The assumption was that a mixture may not manifest clinically as a simple algebraic summation of the biological characteristics of the individual components. From that date, production of new racemates (mixture of enantiomers) ceased to be a rational commercial option and instead became a high-risk route for pharmaceutical companies.
With respect to the enantiomers of clomiphene, it was undoubtedly the differential toxicological profile [2], coupled with relative primary activity as oestrogen antagonists [3], that resulted in the prioritization of Enclomiphene for late stage clinical development. It is this isomer that forms the basis of the submission to the regulatory authorities for the treatment of secondary hypogonadism.
The present paper in some ways reinforces what we already knew; isomers can have differential activity. What is unusual is that what is presented is not pharmacological evidence but clear-cut differences in toxicological profile.
The research raises a specific issue in relation to the treatment of secondary hypogonadism. In general, patients with secondary hypogonadism require restoration of normal testosterone levels while preserving fertility. As such, the use of exogenous testosterone for replacement can be counter-productive as there is a well-documented negative impact on spermatogenesis [4]. As an alternative, ‘off-label’ clomiphene is relatively widely proffered. Assuming approval, Enclomiphene would provide an attractive alternative to its racemic ‘parent’.
Overall, the present paper is a useful reminder of what we think we know; that is, that all racemates are likely to manifest the characteristics of both component enantiomers.
Conflict of Interest
The author is an independent director on the board of Repros Therapeutics.