Editorial: HOXB13 mutations and prostate cancer risk
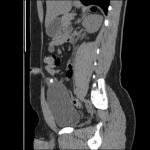
The findings from this study [1], both for the prevalence of the mutation, as well as its magnitude of association with prostate cancer, are comparable to prior reports in Northern European populations. Furthermore, among the 995 cases, the mutation frequency was significantly associated with features predictive of progression after surgery (high PSA level, positive surgical margins, higher pathological Gleason score, and non-organ confined disease) suggesting that genetic evaluation of men with a strong family history would identify a subset of men that would benefit from early screening and intervention in the same manner as are male carriers of known founder mutations in BRCA2[7]. The observation between HOXB13 and clinical features indicative of aggressive disease has been less consistent compared with studies of risk overall and the exact mechanism whereby the gene contributes to malignant progression in the prostate is not well-understood. There is some suggestion that the gene may operate both as a tumour suppressor, as early studies reported its suppression of androgen receptor activity, and as an oncogene as HOXB13 overexpression has been seen in androgen-independent tumours [8].
Currently, most countries (including the USA) do not recommend use of PSA screening for men at average risk for prostate cancer. However, given the significant risk of prostate cancer in men carrying a single copy of the HOXB13 G84E allele, should these male mutation carriers be screened for prostate cancer with PSA testing and DRE? If so, how do we identify these men and at what age should testing commence? Unfortunately, many G84E carriers may not be identified by family history, which raises the question about when is the risk of disease significant enough to warrant population level testing? As Nordic countries, including Denmark, have a higher frequency of HOXB13 G84E allele in the general population, research directed toward understanding the benefit of genetic testing followed by prostate cancer early detection strategies should be considered.